Brno and Göteborg, January 27, 2020
Update 2026: miREIA assay kits have been discontinued as of January 1, 2026.
The microRNA project now continues exclusively on the Two-Tailed RT-qPCR (TT-PCR) platform.
BioVendor licenses Two-tailed PCR technology developed by TATAA Biocenter
Czech company BioVendor with headquarter in Brno licenses Two-Tailed PCR technology for microRNA analysis from Roche. Two-Tailed PCR was developed at TATAA Biocenter in partnership with Roche and is based on an innovative design with an RT primer sensing the microRNA sequence with two linked hemi-probes, which leads to exceeding sensitivity and superior specificity.
Mature microRNAs are single-stranded molecules only 22-24 nucleotides in length. They constitute a recently discovered class of non-coding RNAs that play a key role in the regulation of gene expression. Many microRNAs are present in families that differ only in a single base position or terminal modification. Acting at the post-transcriptional level, microRNAs fine-tune the expression of many mammalian protein-encoding genes. MicroRNAs are present in tissues, but also in biofluids, where they are transported inside exosomes as reporter molecules for uptake at distant locations. This feature makes microRNAs exceedingly attractive biomarkers and its use is currently being standardized by the European effort SPIDIA. There are currently several techniques to measure microRNA levels, but conventional strategies to measure nucleic acids require they are targeted by a pair of primers, each 20-25 bases in lengths. Current methods, therefore, elongate the microRNAs prior to detection, which compromises sensitivity and specificity. The novel Two-tailed approach solves this problem by sensing the microRNA sequence with two short hemiprobes. Neither hemiprobe is long enough to bind alone but in the novel Two-Tailed design the hemiprobes are connected, which leads to cooperative binding, such that either both bind, which gives sufficient contact and stability, or neither binds. Since each hemiprobe is short, a mismatch in its binding site has major impact, leading to the superior specificity of the Two-Tailed PCR method.
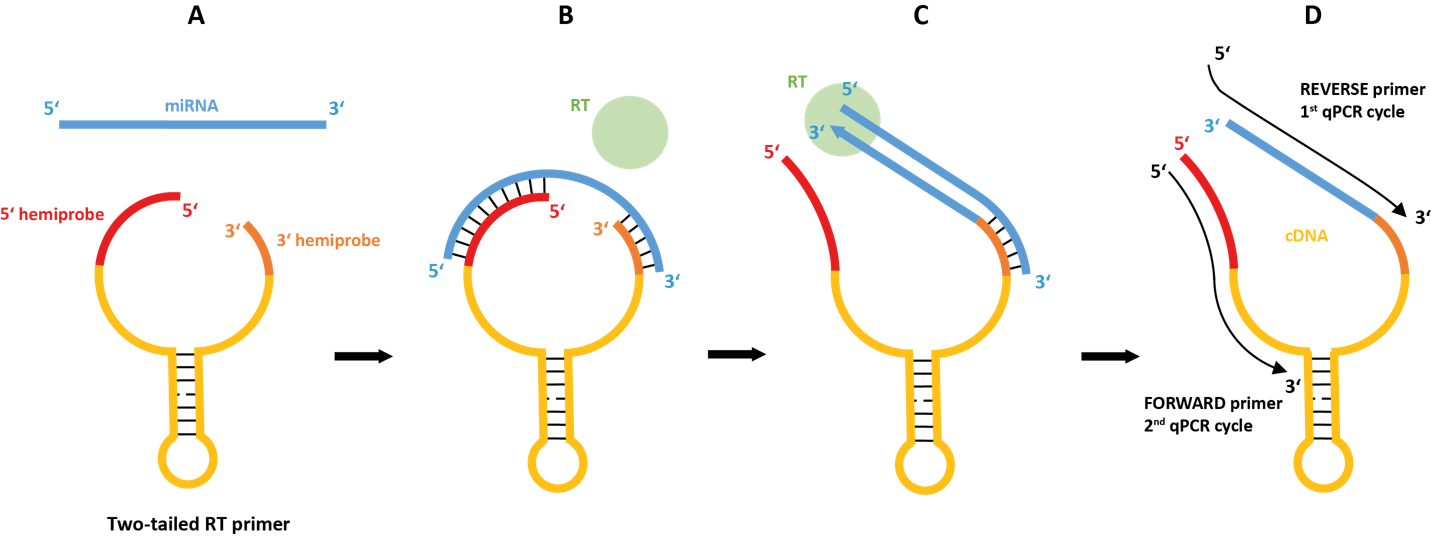
Two-tailed PCR was compared to three other methods for microRNA detection by researchers at the Institute of Biotechnology, BIOCEV and found to be more sensitive and specific, being capable to detect fewer than 10 microRNA molecules in a sample (Nucl. Acids Res., 2017). Two-tailed PCR can be multiplexed to essentially any degree using 2-tube protocol and using one-tube protocol multiplexing with probes is possible. Two-tailed PCR is one of the methods currently evaluated for microRNA detection in liquid biopsies collected for the management of cancer within the European effort Cancer-ID and for Sepsis within the European effort Smartdiagnos. A quality control panel for circulating microRNAs based on Two-Tailed PCR was recently presented (Scientific Reports volume 9, Article number: 4255, 2019). Validated Two-Tailed PCR assays for microRNA analysis are available here.
“We are very glad to partner with BioVendor”, says Mikael Kubista, CEO of TATAA Biocenter. “We have several projects on-going in developing and validating Two-Tailed PCR assays for diagnostics and disease monitoring. BioVendor is a perfect partner joining this work, supporting with expertise on validation and making the panels available to hospitals and researchers globally”.
About TATAA Biocenter
TATAA Biocenter is the world’s largest organizer of hands-on training in quantitative real-time PCR and Next Generation Sequencing and Europe’s leading provider of nucleic acid analysis services. TATAA Biocenter offers a seamless workflow for high-quality analyses of nucleic acids from experimental design, sample preparation, quality control, and data analysis, meeting the specific requirements for compliance with ISO17025 standard. TATAA is particularly known for its focus on quality, contributing to the development of guidelines and standard operating procedures for the diagnostic and pharmaceutical industries, and having co-authored the MIQE guidelines teaching researchers how to report real-time PCR data. Founded in 2001, TATAA Biocenter has laboratories in Gothenburg, Sweden, and in Prague, Czech Republic. In 2019 TATAA Biocenter was named “Best Nucleic Acid Analysis Service Provider – Europe” by Global Health & Pharma. For more info, see. www.tataa.com.
About BioVendor
BioVendor Research and Diagnostic Products is a member of internationally operating BioVendor Group and an innovation hub of the company. Since its establishment in 1992, BioVendor has placed high importance on originality and value brought to customers via providing diagnostics solutions. Recent examples include, fully automated ELISA platform, proprietary microarray solution, Next Generation Sequencing kits, and proprietary miRNA immunoassays. microRNAs have been revealed as an exciting new class of biomarkers standing side by side with the traditional protein markers and BioVendor recognized this potential early on. Starting with a license purchased from Siemens, a miRNA enzyme immunoassay named miREIA has been developed in less than twelve months. Now, BioVendor is a provider of a comprehensive solution for miRNA-oriented researchers. In addition to research, BioVendor is enabling the miRNA biomarkers to enter clinical diagnostic applications. The highly specific and sensitive Two-Tailed PCR technology provides BioVendor with rock-solid base to establish own diagnostic miRNA panels, broaden the life science portfolio, and kickstart BioVendor's rapid expansion in the miRNA market. For more info, see www.biovendor.com.
See also:
- Two-tailed RT-qPCR: a novel method for highly accurate miRNA quantification
- Two-tailed RT-qPCR panel for quality control of circulating microRNA studies
- SPIDIA
- CANCER-ID
- SMARTDIAGNOS
- BTU, BIOCEV
DOWNLOAD THE FLYER
