Description
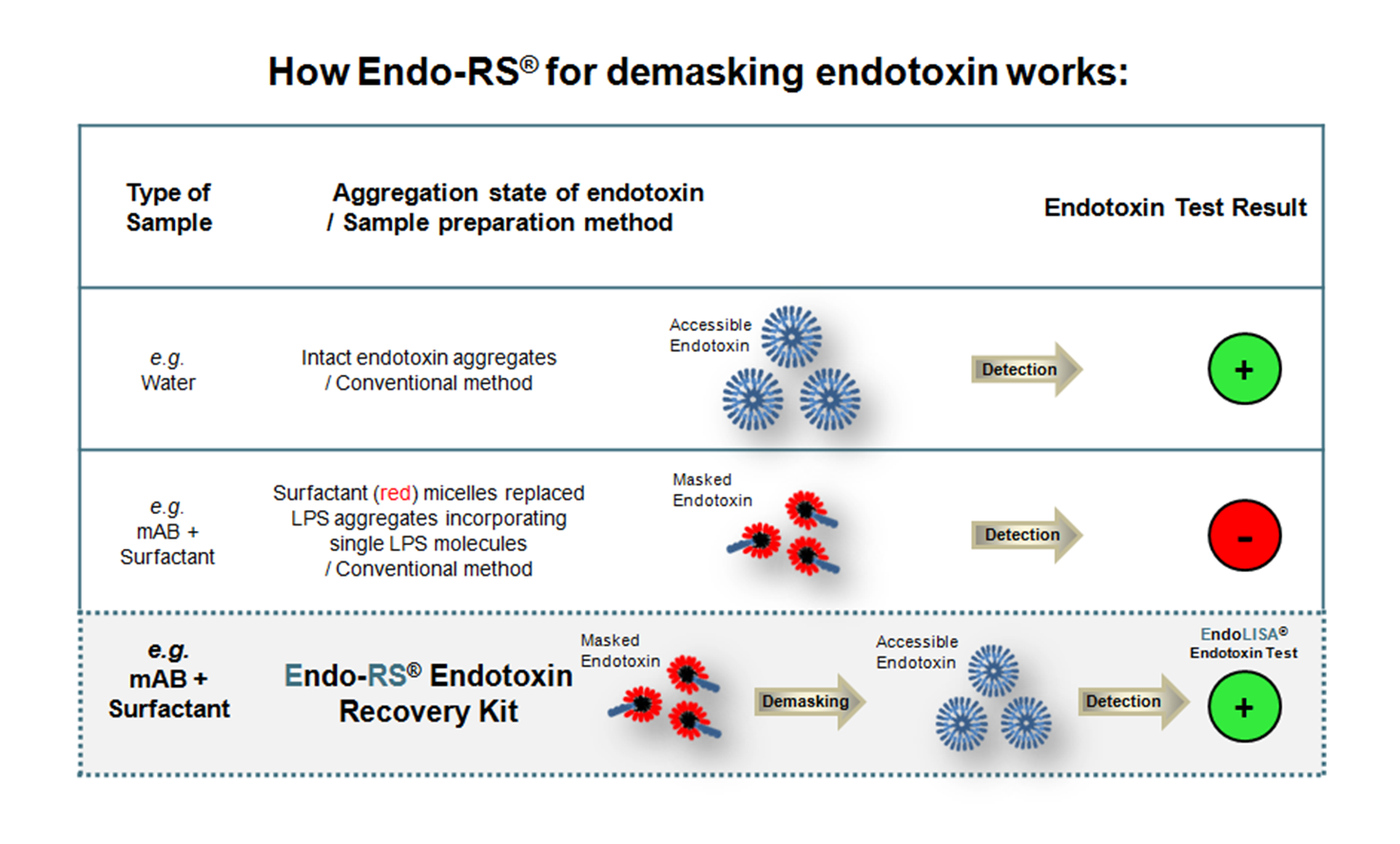
Endo-RS® is a novel sample preparation method enabling complete endotoxin recovery in biopharmaceutical drug formulations containing non-ionic surfactants such as Polysorbate 20, Polysorbate 80 or Octylphenol ethoxylates. Non-ionic surfactants are well known to cause Low Endotoxin Recovery (LER). Intensive studies of the phenomenon have shown that the physicochemical properties of surfactants render endotoxin molecules inaccessible (masked) for detection in such samples. The Endo-RS® Endotoxin Recovery Kit provides the essential reagents and a detailed guideline for evaluating and establishing an optimized endotoxin demasking protocol for each individual formulation containing surfactants. Applying the Endo-RS® Endotoxin Recovery Kit prior to endotoxin detection with the EndoLISA® Endotoxin Detection Assay will ensure FDA-compliant and accurate results in spike validation studies and routine quality control testing.
Shipping
At ambient temperature. Upon receipt, store the product at the temperature recommended below.
Storage/Expiration
Unopened kits are stable at 2–8°C until the expiry date printed on the label.