Cat # changed from RMEE04R to E04
Type
Sandwich ELISA, Biotin-labelled antibody
Applications
Serum, Plasma-EDTA, Plasma-Heparin
Sample Requirements
10 µl/well
Shipping
At ambient temperature. Upon receipt, store the product at the temperature recommended below.
Storage/Expiration
Store the complete kit at 2–8°C. Under these conditions, the kit is stable until the expiration date (see label on the box).
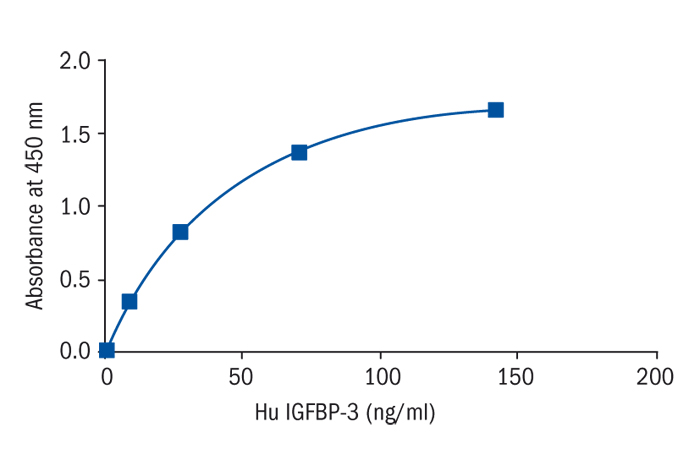
Calibration Curve
Calibration Range
0.4–30 ng/ml
Limit of Detection
0.18 ng/ml
Research topic
Growth hormone and factor-related products
Summary
Insulin-like growth factors (IGF)-1 and -2 are bound to specific binding proteins (IGFBPs) in the circulation. To date, at least six binding proteins can be distinguished on the basis of their amino acid sequence. They are designated as IGFBP-1, IGFBP-2, IGPBP-6. Lately the discovery of a new IGFBP-7 has been discussed. The predominating IGFBP in blood is IGFBP-3. In contrast to the other binding proteins, IGFBP-3 has the unique property to associate with an acid-labile non-binding subunit (ALS) after binding of either IGF-I or IGF-II. Most of the IGFBP-3 in plasma is present as the high molecular weight ternary complex, however, small amounts of free IGFBP-3 are also found.
The development of specific immunoassays for IGFBP-3, those also recognizing the complete high molecular weight complex, provided new in-sights into its regulation (6-9). On the basis of these findings total serum IGFBP-3 has proved to be an additional useful test in the repertoire of diagnostic tools for evaluation of growth disorders. Patients with GH deficiency have subnormal total IGFBP-3 levels. In contrast, most of the small statured children with normal GH secretion have levels within the normal range.
The separation of these two groups is easy. A single measurement of the total IGFBP-3 concentration is sufficient for the diagnosis of GH deficiency with high accuracy. In small statured children total IGFBP-3 levels rise to normal range within several days of GH administration and remain normal during continuous GH treatment. Therefore, total serum IGFBP-3 measurements are also suited for evaluating the potential of a patient to respond to GH and for GH therapy monitoring. In other patients of severe short stature, e.g. Ullrich-Turner syndrome or Silver-Russell syndrome, IGFBP-3 levels were found normal reflecting normal GH secretion. In normal tall children and adolescents without excessive GH secretion or in patients with Sotos syndrome, total IGFBP-3 levels are normal or slightly increased. In contrast, children with pituitary gigantism or adults with acromegaly have clearly elevated levels that normalize on successful treatment. Therefore, total IGFBP-3 is also a useful parameter for the detection of excessive GH secretion and monitoring therapy efficacy. In precocious puberty, total IGFBP-3 levels are clearly increased by chronological age, whereas patients with premature thelarche have total IGFBP-3 levels in the upper normal range. Several factors besides GH influence total IGFBP-3 levels: age including sexual development, nutrition, hypothyroidism, diabetes mellitus, liver function and kidney function. Total IGFBP-3 levels are decreased by malnutrition, although less than IGF-I, in hypothyroidism, in diabetes mellitus and in hepatic failure, but are obviously increased in chronic renal failure. Measurement over 24 hours revealed constant circadian levels. For clinical practice, the most important regulatory factor is GH. Single total IGFBP-3 measurements correlate significantly with the logarithm of the integrated spontaneous GH secretion. In patients with GH deficiency, total IGFBP-3 levels are subnormal and increase gradually to within the normal range after several days of GH administration. The slow response to GH and constant circadian levels during chronic daily application of GH suggest that IGFBP-3 reflects the GH secretory state over days.
Find documents for the lot
Example Instructions for Use (RUO)
Example Instructions for Use (RUO)
Safety Information (RUO)
MSDS (RUO)
MSDS (RUO)