Type
Sandwich ELISA, Biotin-labelled antibody
Applications
Serum, Plasma-EDTA, Plasma-Heparin, Plasma-Citrate, Urine
Sample Requirements
2×5 µl/well
Shipping
At ambient temperature. Upon receipt, store the product at the temperature recommended below.
Storage/Expiration
Store the kit at 2–8°C. Under these conditions, the complete kit is stable until the expiration date (see label on the box).
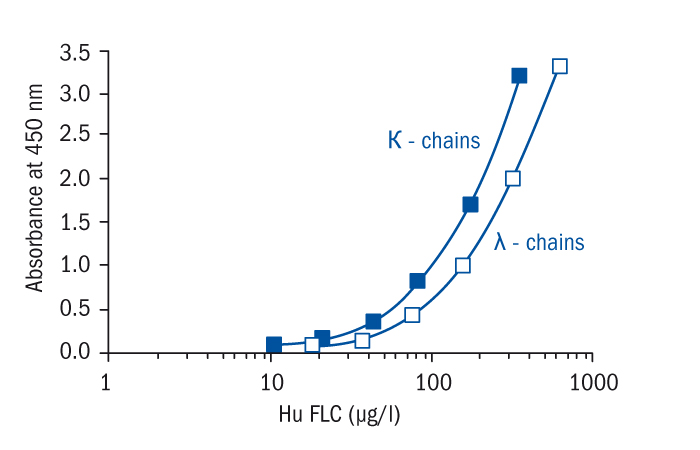
Calibration Curve
Calibration Range
10–320 µg/l (FLC kappa)
17.5–560 µg/l (FLC lambda)
Limit of Detection
FLC kappa ELISA Limit of detection (LOD) (defined as concentration of analyte giving absorbance higher than mean absorbance of blank plus three standard deviations of the absorbance of blank: Ablank + 3×SDblank) is calculated from the real FLC kappa values in wells and is 6 μg/L.
FLC lambda ELISA Limit of detection (LOD) (defined as concentration of analyte giving absorbance higher than mean absorbance of blank plus three standard deviations of the absorbance of blank: Ablank + 3×SDblank) is calculated from the real FLC lambda values in wells and is 5 μg/L.
Intra-assay (Within-Run)
n = 8;
CV = 3.0% (FLC kappa)
CV = 4.5% (FLC lambda)
Inter-assay (Run-to-Run)
n = 6;
CV = 7.0% (FLC kappa)
CV = 6.4% (FLC lambda)
Spiking Recovery
101.4% (FLC kappa)
100.9% (FLC lambda)
Dilution Linearity
87.8% (FLC kappa)
85.8% (FLC lambda)
Crossreactivity
- bovine Non-detectable
- cat Not tested
- goat Non-detectable
- horse Non-detectable
- dog Not tested
- hamster Not tested
- mouse Non-detectable
- pig Non-detectable
- rabbit Non-detectable
- rat Non-detectable
- sheep Non-detectable
- chicken Not tested
- monkey Not tested
- human Yes