Type
Sandwich ELISA, HRP-labelled antibody
Description
Immunoenzymetric assay for the in vitro quantitative measurement of human Intact Parathyroid
Hormone (PTH) in serum and plasma.
Applications
Serum, Plasma
Sample Requirements
200 µl/well
Shipping
On blue ice packs. Upon receipt, store the product at the temperature recommended below.
Storage/Expiration
Store the complete kit at 2–8°C. Under these conditions, the kit is stable until the expiration date (see label on the box).
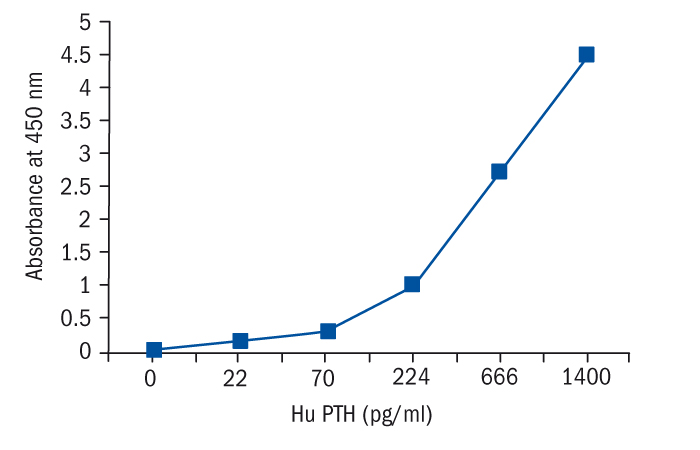
Calibration Curve
Calibration Range
22–1400 pg/ml
Limit of Detection
0.8 pg/ml
Intra-assay (Within-Run)
CV < 4% (serum)
Inter-assay (Run-to-Run)
CV < 5% (serum)
Spiking Recovery
serum 95%, heparin plasma 96%, EDTA plasma 100%
Features
- RUO
- assay for the in vitro quantitative measurement of human Intact Parathyroid
Hormone (PTH) in serum and plasma
- limit of detection 0.8 pg/ml
Research topic
Bone and cartilage metabolism, Oncology
Summary
Human parathyroid hormone (hPTH) is a major physiological regulator of phosphocalcic
metabolism. hPTH increases serum calcium concentrations by its actions on kidney (enhancing
tubular Ca++ reabsorption and phosphate excretion) and bone (stimulating osteoclastic activity
and bone resorption). It indirectly affects intestinal absorption of Ca++ by stimulating renal 1αhydroxylation of 25 hydroxyvitamin D. The release of PTH is controlled in a negative feedback
loop by the serum concentration of Ca++.
PTH is synthesized in the chief cells of the parathyroid glands and secreted as an 84 amino acid
molecule called "intact PTH", which is the main bioactive product. This molecule is degraded by
proteolytic cleavage between amino acids 33-37 at peripheral sites to form biologically active amino- terminal fragments and biologically inactive carboxyl-terminal fragments. The carboxylterminal fragments are cleared only by glomerular filtration, while the bioactive intact PTH and amino-terminal fragments are also metabolically degraded in the liver and other tissues. The halflife of the carboxyl- terminal fragments increases dramatically in patients with renal failure. Thus,
the measurement of intact PTH correlates best with the hormone production and biological activity.
Clinical application
The measurement of intact hPTH is used to establish the diagnosis of primary
hyperparathyroidism by demonstrating elevated serum levels of bioactive PTH. It allows
documenting the occurrence of secondary hyperparathyroidism in patients with Vit.D deficiency,
intestinal malabsorption, or renal failure. In this last situation, the absence of interference with the
inactive carboxyl-terminal fragments is especially valuable. The specificity and high sensitivity of
the assay also allows differentiating clearly low serum PTH levels in hypoparathyroidism or in
tumor-induced hypercalcaemia.
Find documents for the lot
Example Instructions for Use (RUO)
Example Instructions for Use (RUO)