Type
Sandwich ELISA, HRP-labelled antibody
Applications
Serum, Plasma, Cell culture supernatant
Sample Requirements
20 µl/well
Shipping
At ambient temperature. Upon receipt, store the product at the temperature recommended below.
Storage/Expiration
Store the complete kit at 2–8°C. Under these conditions, the kit is stable until the expiration date (see label on the box).
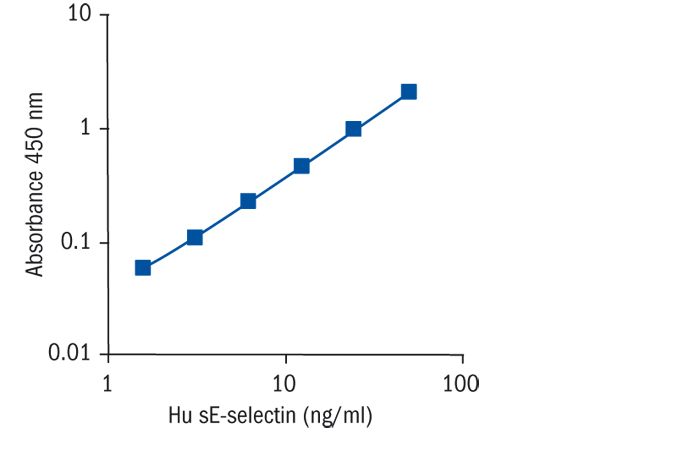
Calibration Curve
Calibration Range
1.6–50 ng/ml
Limit of Detection
0.3 ng/ml
Intra-assay (Within-Run)
CV = 5.4%
Inter-assay (Run-to-Run)
CV = 6.0%
Spiking Recovery
86,00%
Dilution Linearity
95,00%
Research topic
Cell adhesion proteins
Summary
Endothelial Leukocyte Adhesion Molecule-1 (ELAM-1, E-selectin) belongs to the selectin family of adhesion molecules. Together with LECAM-1 (L-selectin) and GMP-140 (P-selectin), Eselectin mediates the initial interactions of leukocytes and platelets with endothelial cells. Molecular structure: The extracellular part of all selectins consists of an aminoterminal c-type lectin domain which specifically binds to carbohydrate ligands. This is followed by an EGF-like domain, and, in the case of E-selectin, by 6 short consensus repeats. The transmembrane portion of the molecule is followed by a short cytoplasmic tail. Selectins guide non-activated polymorphonuclear cells to the areas of inflammation in creating first, loose contacts with the endothelial layer. The potential binding partner of E-selectin contains sialyl LewisX oligosaccharide. Other suitable ligands for the lectin domain of Eselectin are sialylated, fucosylated lactosaminoglycans. Together with GMP-140, E-selectin
is expressed on cytokine-activated endothelial cells, and contributes to the adhesion of still resting leukocytes to the endothelium. This initial binding event is an essential prerequisite for the activation of the immune cells via different inflammatory mediators. In contrast to GMP- 140, E-selectin is maximally expressed 2-4 hours after cell activation. Within the next 24-48 hours E-selectin is again eliminated from the cytoplasmic membrane by shedding into the circulation. The circulating form or soluble (sE-selectin) of this selectin exerts chemotactical signals on neutrophils and additionally activates the 2-integrins - sE-selectin assists in preparing the migration capacity of these cells. Determination of sE-selectin could provide more detailed insights into the pathological
modifications during various diseases:
- Allergic reactions: The transient influx of neutrophils into the respiratory tract, due to an inflammatory response is predominantly mediated via E-selectin. A functional role for this molecule in the development of acute airway inflammation in vivo has been demonstrated. Additionally, E-selectin may be of particular importance for the start phase of allergic contact dermatitis.
- Ocular diseases: The presents of E-selectin on retinal vascular endothelium suggests an important role for this selectin in the pathogenesis of immunologically mediated ocular conditions.
- Septic shock: E-selectin seems to be involved in the pathogenesis of "multiple organ failure (MOF)" during septic shock.
- Vascular infection and inflammation: The levels of sE-selectin in patients with a recent onset of giant cell arteriitis or polyarteriitis nodosa are significantly higher than in normal controls.
- Inflammatory bowel disease: E-selectin is expressed on colonic endothelial surfaces in association with inflammation.
- Transplantation: Increased E-selectin expression on endothelial cells is found in graft-versushost-disease.
Find documents for the lot
Example Instructions for Use (RUO)
Example Instructions for Use (RUO)
Safety Information (RUO)
MSDS (RUO)