Cat # changed from RBCS2000 to S2000
Type
Colorimetric assay
Applications
Cell culture and/or animal studies, Tissue
Shipping
At ambient temperature. Upon receipt, store the product at the temperature recommended below.
Storage/Expiration
All components are stable for one year, (from Invoice Date), when stored at 15–25°C. The glass vial containing collagen standard should be stored at +4°C once opened.
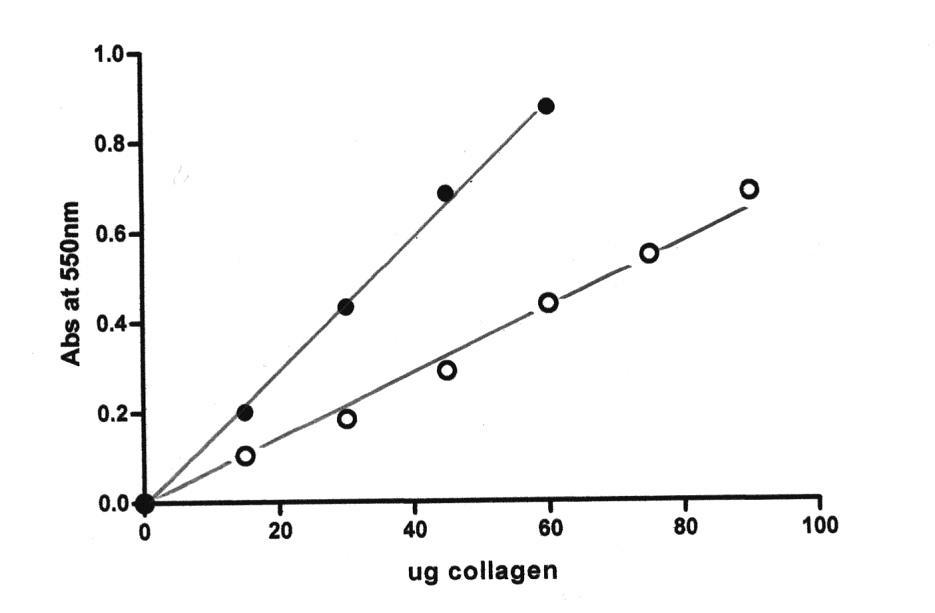
Calibration Curve
Limit of Detection
10 μg
Features
- The Sircol™ INSOLUBLE Collagen Assay is a dye-binding method for analysis of insoluble collagen fibers.
- The assay be used to assess the rate of production of newly laid down collagen fibers during periods of rapid growth, development, tissue repair, remodeling and wound healing. Sources of material includes tissues, bone and calcified tissue.
- It replaces the conventional hydroxyproline analysis. No more working with concentrated acids or alkalis!
- The Sircol Insoluble Assay uses mild acid and temperature treatment for 2 to 3 hrs.
- The solubility conversion to denatured collagen and subsequent measurement can be completed within one working day.
- The ratio of soluble and insoluble collagens can be measured using the same Sircol Dye Reagent.
- Manufactured by Biocolor.
Research topic
Extracellular matrix, Animal studies
Summary
Soluble and Insoluble Collagen measurement Collagen is the most abundant protein found in animals. During a healthy life span the insoluble, covalent, cross-linked collagen fibers retain their biophysical functions and shapes. The collagen fibers remain isolated from most of the biochemical activities of the cells.
Trauma caused by metabolic internal events, external chemical agents or physical injuries however, quickly lead to dramatic activity, resulting in rapid collagen removal followed by a wound healing response (collagen regeneration and associated remodeling). Newly formed soluble collagen production and the residual insoluble collagen fibers can be monitored using the Sircol Soluble Collagen and Sircol Insoluble Collagen Assays.
Collagen proteins contain one or more domains with a triple helical structure. The three chains are described as alpha chains and should not be confused with the alpha helixes found in other proteins. The fibrillar collagens (Types I, II, III, V & XI) have most of their alpha chain structure composed of a continuous repeating tri-peptide sequence made up of glycine in every third amino acid residue [(gly-X-Y)n ]. It is to this sequence that the Sircol Dye binds.
Proline is frequently an occupant in the ‘Y’ location of the tri-peptide [(gly-X-Y)n]. Many of these residues are converted, post translation, into hydroxyproline residues prior to triple helix formation and the release of the tropocollagen into the ECM.
Until now, an investigator seeking to measure covalent cross-linked insoluble collagen required measurements based on free hydroxyproline content - a procedure that requires strong acid (6.0 M HCl), high temperatures (+ 95 °C) and overnight cooking (18 to 24 hrs).