Type
Sandwich ELISA, HRP-labelled antibody
Applications
Serum, Plasma, Cell culture supernatant
Sample Requirements
10 µl/well
Shipping
At ambient temperature. Upon receipt, store the product at the temperature recommended below.
Storage/Expiration
Store the complete kit at 2–8°C. Under these conditions, the kit is stable until the expiration date (see label on the box).
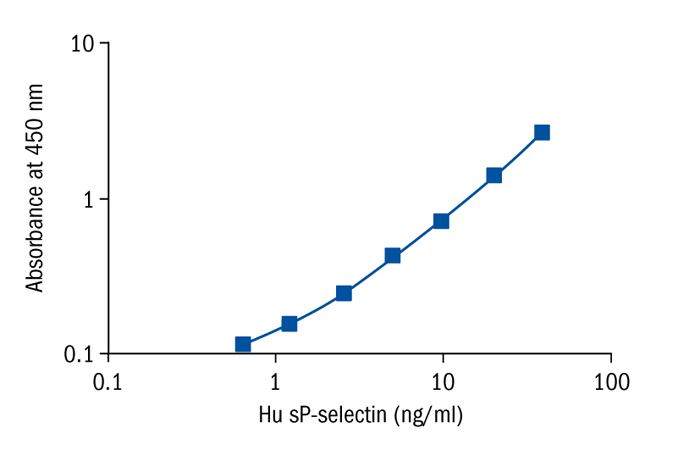
Calibration Curve
Calibration Range
0.63–40.00 ng/ml
Limit of Detection
0.20 ng/ml
Intra-assay (Within-Run)
CV = 7.8%
Inter-assay (Run-to-Run)
CV = 5.4%
Dilution Linearity
91,60%
Research topic
Cell adhesion proteins
Summary
P-selectin (CD62, GMP-140, PADGEM) belongs to the selectin family of adhesion molecules. P-selectin acts as a receptor that supports binding of leukocytes to activated platelets and endothelium. P-selectin-mediated adhesive interactions operate in conjunction with cell-cell interactions directed by related molecules and are likely to be important in both hemostatic and inflammatory processes.
P-selectin is located in membranes of a granules in unstimulated platelets and redistributed to the cell surface upon platelet activation. P-selectin is also present in endothelial cells in membranes of Weibel-Palade bodies and megakaryocytes. Surface appearance of P-selectin is very rapid, but transient declining to basal level within short time following stimulation.
P-selectin is a 140 kDa protein that is highly glycosylated. The cDNA-derived amino acid sequence predicts a molecule with a series of cysteine-rich domains. Like the other selectins P-selectin contains an N-terminal Ca2+ dependent lectin-like domain and an EGF-like motif which is followed by nine consensus repeats, a transmembrane domain, and a short cytoplasmic tail. The human gene for P-selectin is located on chromosome 1q21-24. P-selectin is a receptor for neutrophils and monocytes, recognizing oligosaccharide structures on the target cells. The physiologic role of P-selectin might be the mediation of initial leukocyte adhesion to activated endothelium during acute inflammation. It may work in concert with E-selectin to direct early, regionally specific adherence of neutrophils and monocytes at sites of acute inflammation. A soluble form of P-selectin found in serum and plasma has been described which might
represent a proteolytic fragment or more likely a soluble splice variant lacking the transmembrane domain.
Soluble P-selectin is a potentially important molecule to provide more detailed insight into pathological situations. Excessive accumulation of neutrophils on the endothelial surface accompanied by high exposure of P-selectin has been implicated in a number of inflammatory disorders, including adult respiratory distress syndrome, acute lung injury, ischemiareperfusion injury, Gram-negative septic shock, thrombotic diseases and rheumatoid arthritis. Malignant cells were shown to express receptors for P-selectin suggesting an important role for P-selectin in tumor formation and metastasis. Platelets have also been shown to promote tumor metastasis.
Find documents for the lot
Example Instructions for Use (RUO)
Example Instructions for Use (RUO)
Safety Information (RUO)
MSDS (RUO)