Type
Sandwich ELISA, HRP-labelled antibody
Applications
Serum, Urine, Plasma, Cell culture supernatant
Sample Requirements
10 µl/well
Shipping
At ambient temperature. Upon receipt, store the product at the temperature recommended below.
Storage/Expiration
Store kit reagents between 2–8°C except controls. Store lyophilized controls at -20°C.
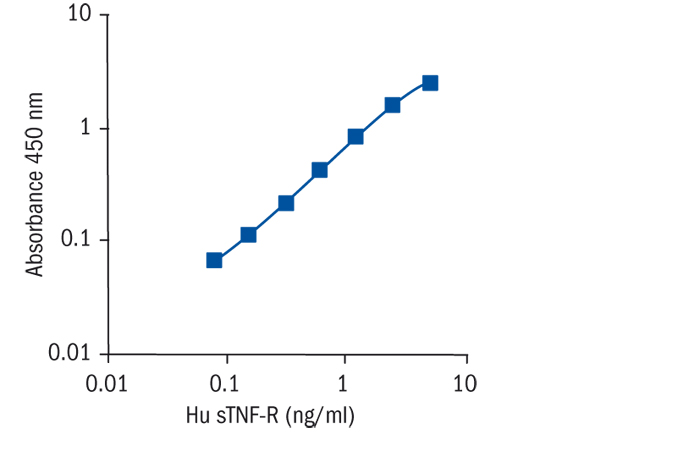
Calibration Curve
Calibration Range
0.08–5 ng/ml
Limit of Detection
0.05 ng/ml
Intra-assay (Within-Run)
CV = 1.9%
Inter-assay (Run-to-Run)
CV = 8.6%
Spiking Recovery
93,00%
Dilution Linearity
103,00%
Research topic
Cytokines and chemokines and related molecules
Summary
Tumor Necrosis Factor (TNF) was originally discovered in sera of animals and was found to cause hemorrhagic necrosis of some transplantable mouse and human tumors and to exhibit primarily cytotoxic activities against tumor but not normal cells in vitro. The TNF family consists of two proteins designated TNFα, also called cachectin, and TNFβ, also called lymphotoxin, which are pleiotropic cytokines that can mediate a wide variety of biological effects.
Both TNFα and TNFβ have been shown to interact with a cell through specific high affinity receptors with a few hundred up to more than 20,000 copies per cell. TNF-receptors have been demonstrated on a wide variety of human somatic cells including fibroblasts, endothelial cells, adipocytes, liver membranes, granulocytes and several tumor cell lines. Normal and malignant human myeloid cells as well as mitogen-stimulated lymphocytes express similar numbers of TNF receptors (400 - 1,900 per cell), whereas resting lymphoid cells have fewer, red blood cells and platelets have no detectable TNF receptors.
In most cases no correlation is observed between receptor number and sensitivity to TNF. Based on gel filtration experiments the receptor appears to be a complex of different proteins with a molecular weight of 350 kDa. In a variety of cell lines two different types of TNF receptors with 75-80 and 55-60 kDa respectively have been identified.
The cDNAs encoding the two different TNF receptors have been cloned. The exact mechanism of signal transduction after binding of TNF to the receptor is still unclear. The extracellular fragment of the 60 kDa TNF receptor, with a molecular mass of about 30 kDa was purified, partially sequenced, and the respective cDNA was cloned. This TNF binding protein is liberated from the intact molecule by proteolytic cleavage and comprises most of the extracellular portion of the receptor, including all three N-glycosylation sites.
The present assay provides a simple, rapid, and highly sensitive method for the determination of soluble TNF-R (60 kDa) levels in body fluids or cell culture supernatants. This assay will help to clarify the possible diagnostic and prognostic value of circulating sTNF-R (60 kDa) in various neoplastic and inflammatory diseases.
Find documents for the lot
Example Instructions for Use (RUO)
Example Instructions for Use (RUO)
Safety Information (RUO)
MSDS (RUO)