Type
Competitive ELISA
Description
The BioVendor Testosterone rat/mouse ELISA is a competitive immunoassay for the measurement of testosterone in rat and mouse serum or plasma (EDTA). For research use only. Not for use in diagnostic procedures.
Applications
Serum, Plasma-EDTA
Sample Requirements
10 µl/well
Shipping
On blue ice packs. Upon receipt, store the product at the temperature recommended below.
Storage/Expiration
Store the complete kit at 2–8°C. Under these conditions, the kit is stable until the expiration date (see label on the box).
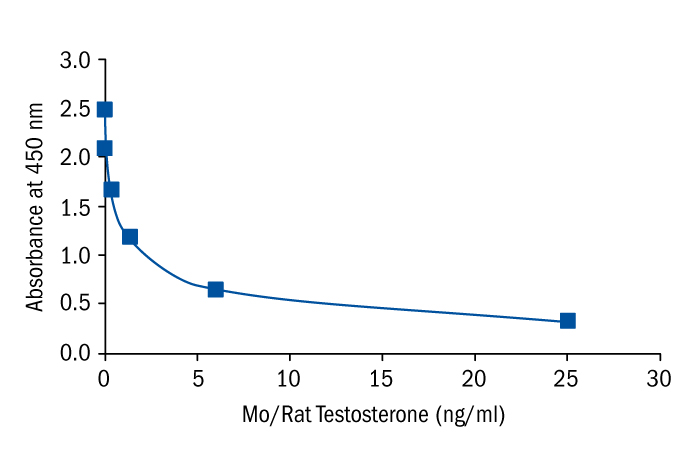
Calibration Curve
Calibration Range
0.1–25 ng/ml
Limit of Detection
0.024 ng/ml
Features
- RUO
- calibration range 0.1-25 ng/ml
- limit od detection 0.024 ng/ml
Research topic
Steroid hormones, Animal studies
Summary
Testosterone is a steroid hormone from the androgen group synthesized by the Leydig cells in the testes in males, the ovaries in females, and adrenal glands in both sexes. It exerts a wide-ranging influence on sexual behaviour, muscle mass and strength, energy, cardiovascular health, and bone integrity.
Testosterone biosynthesis coincides with the spermatogenesis and fetal Leydig cell differentiation in the male rat. Several in vivo models including hormone suppression, hormone restoration and hypophysectomy were established for the study of the hormonal regulation of spermatogenesis by testosterone (1-3).
In the Brown Norway rat, serum testosterone levels decrease with aging, accompanied by increases in serum FSH. The capacity of Leydig cells to produce testosterone is higher in young than in old rats (4). Testosterone secreted during late gestational and neonatal periods causes significant brain sexual dimorphism in the rat. This results in both sex-specific behaviour and endocrinology in adults (5).
Analyses concerning the regulation of synthesis reveal that testosterone is able to regulate its own synthesis and indicate that this autoregulation is the result of rapid, specific inhibition by testosterone of 17 alpha-hydroxylase activity (6).
Find documents for the lot
Example Instructions for Use (RUO)
Example Instructions for Use (RUO)
Safety Information (RUO)
MSDS (RUO)