Type
Recombinant protein
Description
Total 348 AA. UniProtKB acc.no. P43490. N-terminal Flag 11 AA. Mw: 39.6 kDa (calculated).
Amino Acid Sequence
MDYKDDDDKASPPNTSKVYSYFECREKKTENSKLRKVKYEETVFYGLQYILNKYLKGKVVTKEKIQEAKDVYKEHFQDDVFNEKGWNYILEKYDGHLPIEIKAVPEGFVIPRGNVLFTVENTDPECYWLTNWIETILVQSWYPITVATNSREQKKILAKYLLETSGNLDGLEYKLHDFGYRGVSSQETAGIGASAHLVNFKGTDTVAGLALIKKYYGTKDPVPGYSVPAAEHSTITAWGKDHEKDAFEHIVTQFSSVPVSVVSDSYDIYNACEKIWGEDLRHLIVSRSTQAPLIIRPDSGNPLDTVLKVLEILGKKFPVTENSKGYKLLPPYLRVIQGDGVDINTLQE
Source
E. coli
Purity
˃ 90 % by SDS-PAGE
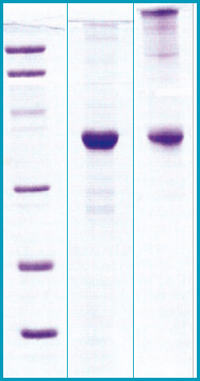
SDS-PAGE Gel
12% SDS-PAGE separation of Human Visfatin
1. M.W. marker – 14, 21, 31, 45, 66, 97 kDa
2. reduced and heated sample, 5μg/lane
3. non-reduced and non-heated sample, 5μg/lane
Endotoxin
< 1.0 EU/µg
Formulation
Filtered (0.4 μm) and lyophilized in 20 mM Tris buffer, 50 mM NaCl, pH 7.5
Reconstitution
Add deionized water to prepare a working stock solution of approximately 0.5 mg/mL and let the lyophilized pellet dissolve completely.
Applications
Western blotting
Shipping
At ambient temperature. Upon receipt, store the product at the temperature recommended below.
Storage/Expiration
Store the lyophilized protein at -80 °C. Lyophilized protein remains stable until the expiry date when stored at -80 °C. Aliquot reconstituted protein to avoid repeated freezing/thawing cycles and store at -80 °C for long term storage. Reconstituted protein can be stored at 4 °C for a week.
Quality Control Test
BCA to determine quantity of the protein.
SDS PAGE to determine purity of the protein.
Endotoxin level determination.
Note
This product is intended for research use only.
Research topic
Cytokines and chemokines and related molecules, Energy metabolism and body weight regulation
Summary
Excess adiposity is the most important risk in the development of insulin resistance and type 2 diabetes mellitus (T2DM). Adipose tissue produces several proteins (adipocytokines) such as leptin, adiponectin, resistin, tumor necrosis factor-α, and IL-6, that modulate insulin sensitivity and appear to play an important role in the pathogenesis of insulin resistance, diabetes, dyslipidemia, inflammation, and atherosclerosis. However, the mechanisms by which fat tissue induces insulin resistance and the role of adipocytokines in the pathogenesis of T2DM have not been well established. Visfatin, also known as pre-B cell colony-enhancing factor (PBEF), is a cytokine that is highly expressed in visceral fat and was originally isolated as a secreted factor that synergizes with IL-7 and stem cell factors to promote the growth of B cell precursors. Visfatin homologs have been identified in carp, invertebrate mollusks , and bacteria, as well as in vertebrates, including humans and the mouse. It has been postulated to play a role in innate immunity. Visfatin exerts insulin-mimetic effects that are dose-dependent and quantitatively similar to those of insulin in stimulating muscle and adipocyte glucose transport, and in inhibiting hepatocyte glucose production. Intravenous injection of recombinant visfatin in mice decreased plasma glucose in a dose-dependent fashion. In keeping with its insulin-mimetic effects, visfatin was as effective as insulin in reducing hyperglycemia in insulin-deficient diabetic mice. Visfatin was also found to be bound to and activate insulin receptor, causing receptor phosphorylation and the activation of downstream signaling molecules. However, visfatin and insulin did not compete for binding to the insulin receptor, indicating that the two proteins were recognized by different regions of the receptor. Thus, visfatin might play a role in glucose homeostasis and dysregulation in biosynthesis or signal transduction, and might contribute to the pathogenesis of diabetes.