Type
Sandwich ELISA, HRP-labelled antibody
Description
The RIS001R Human Aggrecan (PG) ELISA Immunoenzymetric assay for the measurement
of human aggrecan (PG) in synovial fluid, serum and cell culture supernatant. It is intended
for research use only.
Applications
Serum, Synovial fluid, Cell culture supernatant
Sample Requirements
50 µl/well
Shipping
On blue ice packs. Upon receipt, store the product at the temperature recommended below.
Storage/Expiration
Store the complete kit at 2–8°C. Under these conditions, the kit is stable until the expiration date (see label on the box).
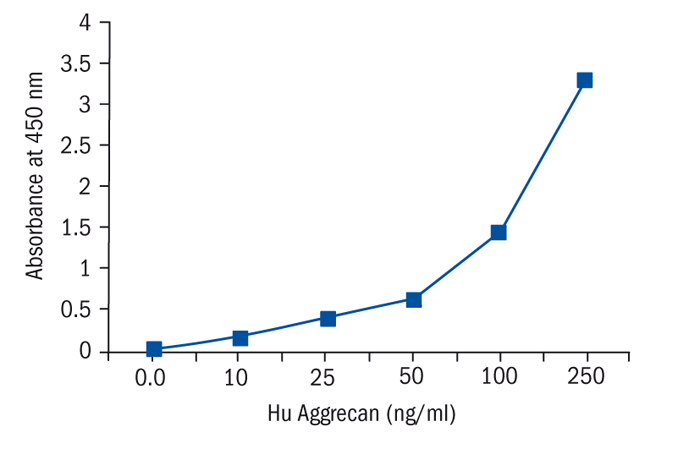
Calibration Curve
Calibration Range
10–250 ng/ml
Limit of Detection
0.9 ng/ml
Features
- RUO
- calibration range 10-250 ng/ml
- limit of detection 0.9 ng/ml
- assay for the measurement of human aggrecan (PG) in synovial fluid, serum and cell culture supernatant
- lyophilized controls
Research topic
Bone and cartilage metabolism, Extracellular matrix
Summary
Aggrecan (PG) is the predominant proteoglycan species in articular cartilage. It is composed of a
core protein of 210 kDa to which over 100 chondroitin sulfate chains, about 20-50 keratan sulfate
chains and O-linked as well as N-linked oligosaccharides are covently attached. The core protein
contains three distinct globular domains (G1-G3). G1 is at the amino terminus, separated by a
short extended segment from G2, while G3 is at the carboxy terminal end. The G1 amino terminal
region can interact noncovalently with hyaluronic acid (HA) and has then be termed the hyaluronic
acid binding region (HABR). A link protein interact with both the G1 region and the HA to stabilize
this interaction. PG is produced by chondrocytes, and its production is regulated by cytokines and
growth factors such as IL1β, TNFα, IGF1 or TGFβ. In extracellular matrix, as many as 200
aggrecan molecules can bind to one single HA molecule to form an aggregate (MW: 5.107
to 5.108).
An imbalance in the synthesis and degradation of the matrix components is a common feature of
both osteoarthritis and rheumatoid arthritis. The loss of PG and other matrix components from the cartilage leads to destruction of the tissue, causing complete deterioration of the articular surface.
Several recent publications suggest that the PG and PG fragments released in synovial fluid and
serum during the degradation process might serve as markers of the metabolic changes in
diseased cartilage. Cell culture is a commonly used procedure for the study of cartilage
metabolism. The measurement of PG and other matrix components in culture supernatants and
cellular contents can assist analysis of the effects of cytokines, growth factors, drugs and potential
chondroprotective substances on the cartilage homeostasy. Biovendor has developped a ELISA
for the measurement of human aggrecan to aid the study of this important cartilage constituent.
The PG-ELISA is convenient, highly specific, and allow accurate measurement of PG in synovial
fluid, serum and culture supernatant This assay is a new interesting tool for the exploration of the
cartilage metabolism.
Find documents for the lot
Example Instructions for Use (RUO)
Example Instructions for Use (RUO)
Safety Information (RUO)
MSDS (RUO)