Type
Recombinant protein
Description
Total 392 AA. MW: 44.2 kDa (calculated). UniProtKB/Swiss-Prot Q9BY76 with C-Terminal Flag-tag, 11 extra AA.
Amino Acid Sequence
GPVQSKSPRFASWDEMNVLAHGLLQLGQGLREHAERTRSQLSALERRLSACGSACQGTEGSTDLPLAPESRVDPEVLHSLQTQLKAQNSRIQQLFHKVAQQQRHLEKQHLRIQHLQSQFGLLDHKHLDHEVAKPARRKRLPEMAQPVDPAHNVSRLHRLPRDCQELFQVGERQSGLFEIQPQGSPPFLVNCKMTSDGGWTVIQRRHDGSVDFNRPWEAYKAGFGDPHGEFWLGLEKVHSITGDRNSRLAVQLRDWDGNAELLQFSVHLGGEDTAYSLQLTAPVAGQLGATTVPPSGLSVPFSTWDQDHDLRRDKNCAKSLSGGWWFGTCSHSNLNGQYFRSIPQQRQKLKKGIFWKTWRGRYYPLQATTMLIQPMAAEAASAAADYKDDDDK
Source
HEK293
Purity
˃ 90 % by SDS-PAGE
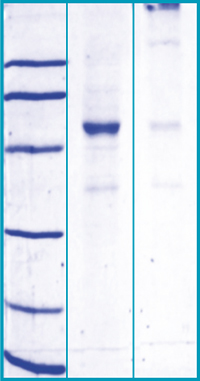
SDS-PAGE Gel
12% SDS-PAGE separation of Human ANGPTL4
1. M.W. marker – 14, 21, 31, 45, 66, 97 kDa
2. reduced and heated sample, 10μg/lane
3. non-reduced and non-heated sample,10μg/lane
Endotoxin
< 1.0 EU/µg
Formulation
Filtered (0.4 μm) and lyophilized in 0.5 mg/mL in 0.05 M phosphate buffer, 0.075 M NaCl, pH 7.4
Reconstitution
Add deionized water to prepare a working stock solution of approximately 0.5 mg/mL and let the lyophilized pellet dissolve completely.
Applications
Western blotting, ELISA
Shipping
At ambient temperature. Upon receipt, store the product at the temperature recommended below.
Storage/Expiration
Store the lyophilized protein at -80 °C. Lyophilized protein remains stable until the expiry date when stored at -80 °C. Aliquot reconstituted protein to avoid repeated freezing/thawing cycles and store at -80 °C for long term storage. Reconstituted protein can be stored at 4 °C for a week.
Quality Control Test
BCA to determine quantity of the protein.
SDS PAGE to determine purity of the protein.
Endotoxin level determination.
Note
This product is intended for research use only.
Research topic
Energy metabolism and body weight regulation
Summary
Angiopoietin-like protein 4 (ANGPTL4) is a secreted 50 kD protein that modulates the disposition of circulating triglycerides (TG) by inhibiting lipoprotein lipase (LPL). ANGPTL4 was identified as a gene that is induced by fasting, and during 3T3-L1 preadipocyte differentiation, and was thus named HFARP (hepatic fibrinogen/angiopoietin-related protein), FIAF (fasting-induced adipose factor), and PGAR (PPAR-angiopoietin related). It is one of the seven members of the angiopoietin-like family. Angptl4 is expressed ubiquitously, predominantly in adipose tissue, liver, placenta, myocardium, keratinocytes, podocytes, intestine, and pituitary gland. ANGPTL4 is a fusion protein consisting of an N-terminal coiled-coil domain and a C-terminal fibrinogen-like domain. These two domains have been shown to have distinct biological functions. The N-terminal domain is responsible for the inhibitory effects on LPL, converting the active form of LPL into an inactive form, and the C-terminus mediates its antiangiogenic functions. Interestingly, these two domains are separated by a short linker that can be cleaved after secretion. Upon secretion into the circulation, ANGPTL4 is cleaved into an N-terminal domain and a C-terminal fibrinogen-like domain. The N-terminal peptide circulates as an oligomer, and the fibrinogen-like domain circulates as a monomer. The N-terminal domain of ANGPTL4 interacts directly but transiently with LPL, triggering a stable conformational switch in LPL that irreversibly inactivates the enzyme. Cleavage of ANGPTL4 appears to be tissue-dependent in humans; liver secretes cleaved ANGPTL4, whereas adipose tissue secretes the full-length form. In mice the full-length form of ANGPTL4 is physically associated with HDL, whereas truncated ANGPTL4 is associated with low density lipoprotein. In humans, both full-length and truncated ANGPTL4 are associated with HDL. ANGPTL4 expression is upreguated by fasting, free fatty acids, PARR agonists, acute phase response, glucocorticoids, and downregulated by insulin. ANGPTL4 has been implicated in a variety of diseases, including cardiovascular disease, cancer metastasis, obesity, diabetes, wound repair, inflammation, arthritis and nephrotic syndrome. Serum or plasma levels were determined in a limited number of studies. ANGPTL4 serum levels display high variability between individuals ranging from 2 to 158 ng/ml. In post-heparin plasma, ANGPTL4 is increased. ANGPTL4 correlates positively with age, body fat mass, waist-hip-ratio and free faty acids but negatively with plasma high-density lipoprotein cholesterol. No correlation with triglycerides was observed in one study. ANGPTL4 is a positive acute phase protein and its increase could contribute to the hypertriglyceridemia that characteristically occurs during the acute phase response by inhibiting LPL activity.