Type
Recombinant protein
Description
Total 193 AA. MW: 22 kDa (calculated). UniProtKB acc.no. P23284 (Asp34-Glu216). N-terminal His-tag (10 extra AA). Protein identity confirmed by MS.
Amino Acid Sequence
MKHHHHHHASDEKKKGPKVTVKVYFDLRIGDEDVGRVIFGLFGKTVPKTVDNFVALATGEKGFGYKNSKFHRVIKDFMIQGGDFTRGDGTGGKSIYGERFPDENFKLKHYGPGWVSMANAGKDTNGSQFFITTVKTAWLDGKHVVFGKVLEGMEVVRKVESTKTDSRDKPLKDVIIADCGKIEVEKPFAIAKE
Source
E. coli
Purity
˃ 90 % by SDS-PAGE
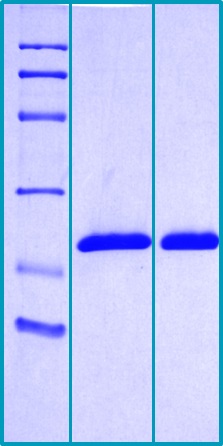
SDS-PAGE Gel
14 % SDS-PAGE separation of Human Cyclophilin B:
1. M.W. marker – 14, 21, 31, 45, 66, 97 kDa
2. reduced and boiled sample, 2.5 μg/lane
3. non-reduced and non-boiled sample, 2.5 μg/lane
Endotoxin
< 1.0 EU/µg
Formulation
Filtered (0.4 μm) and lyophilized from 0.5 mg/mL solution in 20 mM Tris buffer, 50 mM NaCl, pH 7.5.
Reconstitution
Add 200 µl of deionized water to prepare a working stock solution of approximately 0.5 mg/ml and let the lyophilized pellet dissolve completely.
Applications
Western blotting, ELISA
Shipping
At ambient temperature. Upon receipt, store the product at the temperature recommended below.
Storage/Expiration
Store the lyophilized protein at -80 °C. Lyophilized protein remains stable until the expiry date when stored at -80 °C. Aliquot reconstituted protein to avoid repeated freezing/thawing cycles and store at -80 °C for long term storage. Reconstituted protein can be stored at 4 °C for three days.
Quality Control Test
BCA to determine quantity of the protein.
SDS PAGE to determine purity of the protein.
Endotoxin level determination.
Note
This product is intended for research use only.
Research topic
Immunology, Oncology
Summary
Cyclophilin B (CypB, Peptidyl-prolyl cis-trans isomerase B, PPIase B, CYP-S1, rotamerase B, S-cyclophilin, SCYLP, PPIB) is a 21-kDa protein belonging to the cyclophilin family. Cyclophilins are ubiquitous cellular proteins with peptidyl-prolyl cis-trans isomerase enzymatic activity, which are involved in a variety of functions related to cell metabolism, energy homeostasis, and exhibit enhanced expression in inflammation or malignancy.
CypB has been identified in the endoplasmic reticulum and nucleus of all cell types and is also secreted in notable levels in the serum and breast milk. Structurally, CypB shows a high degree of homology with other members of the cyclophilin family in its core β-barrel/isomerase region, which contains a surface hydrophobic pocket that constitutes the proline binding motif. Both N- and C- termini of CypB differ significantly from other cyclophilin family members. Within the N-terminus, CypB contains a nuclear translocation motif (DEKKKGPKV), while the endoplasmic reticulum retention sequence (AIAKE) resides in its C-terminus. This ER-retention motif is proteolytically clipped in the ER, enabling secretion of CypB into the extracellular milieu.
The larger biological function of CypB has remained enigmatic. Within the nucleus, ongoing research has demonstrated that CypB functions as a transcriptional inducer of Stat5-mediated gene expression. Given the association of CypB with other intracellular transcription factors, these findings suggest that CypB could serve to coordinate global networks of gene expression. At the cell surface, CypB also serves as a ligand for CD147 receptor that in turn regulates mitogen-activated protein kinase activation, motility, calcium transport and expression of the pro-apoptotic protein Bim.
The extracellular fractions of CypB are involved in cell-cell communication and inflammatory signaling however on its own, CypB seems to be unable to induce pro-inflammatory cytokines. CypB acts in progression of inflammatory diseases such as rheumatoid arthritis and psoriasis, but is equally involved in the first steps of certain viral infections. Its inflammatory activity is conditioned by its interaction with heparan sulfate proteoglycans and the membrane receptor CD147, two binding partners at the cell surface of T cell lymphocytes, granulocytes and macrophages. CypB is able to induce chemotaxis and triggers T lymphocyte adhesion to fibronectin in the extracellular matrix.
Cyclophilin B is closely associated with the occurrence and progression of tumors. This protein has been shown to be highly expressed in breast, liver, colon, stomach and pancreatic cancer.