Type
Sandwich ELISA, Biotin-labelled antibody
Description
The human IL-33 ELISA is an enzyme-linked immunosorbent assay for the quantitative detection of human IL-33. The human IL-33 ELISA is for research use only. Not for diagnostic or therapeutic procedures.
Applications
Serum, Plasma-Heparin, Plasma-Citrate, Cell culture supernatant
Sample Requirements
50 µl/well
Shipping
On blue ice packs. Upon receipt, store the product at the temperature recommended below.
Storage/Expiration
Store the complete kit at 2–8°C. Under these conditions, the kit is stable until the expiration date (see label on the box).
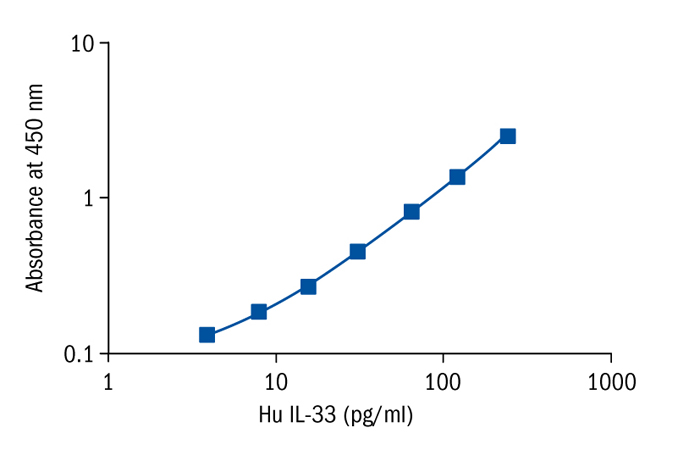
Calibration Curve
Calibration Range
7.8–500 pg/ml
Limit of Detection
0.9 pg/ml
Intra-assay (Within-Run)
CV = 4.7%
Inter-assay (Run-to-Run)
CV = 6.9%
Dilution Linearity
99%
Specificity
The assay detects both natural and recombinant human IL-33.
The cross reactivity and interference of circulating factors of the immune system was evaluated by spiking these proteins at physiologically relevant concentrations into a human IL-33 positive sample.
There was no cross reactivity or interference detected, notably not with IL-1α, IL-1β and IL-18.
Features
- RUO
- calibration range 7.8-500 pg/ml
- limit of detection 0.9 pg/ml
- intra-assay CV= 4.7%
- inter-assay CV = 6.9%
Research topic
Cardiovascular disease, Cytokines and chemokines and related molecules, Energy metabolism and body weight regulation, Oncology
Summary
IL-33 is the 11th and most recently discovered member of the IL-1 family of cytokines, which furthermore includes IL-1α, IL-1β, IL-18 and IL-1Ra. As with IL-1β and IL-18, the synthesized 30 kDa propeptide IL-33 lacks a clear signal peptide for direct processing via the endoplasmic reticulum and Golgi apparatus. In vivo, caspase-1 cleaves the pro-IL-1β and pro-IL-18 bioactive forms, an essential step for their subsequent secretion. The mechanism for IL-33 might be similar, but the process has not been fully elucidated yet. In vitro, caspase-1 can
cleave human IL-33, which results in a 20-22 kDa mature form. In humans, IL-33 mRNA is predominantly found in dermal fibroblasts, bronchial and small airway epithelial cells and smooth muscle cells of skin and lung tissues. Later, cellular mRNA expression has been observed in adipocytes, synovial fibroblasts, high endothelial venules, and endothelial cells. Furthermore, IL-33 is expressed in fibroblastic reticular cells of lymphoid tissues, skin keratinocytes, epithelial cells of stomach, tonsillar crypts and salivary glands, cardiac
fibroblasts and cardiomyocytes. In most cells the predominant localization of IL-33 is nuclear rather than cytoplasmic. IL-33 specifically binds to IL1RL-1, which is known also as ST2, and is part of the IL-1R family. IL-33 promotes the release of Th2-associated cytokines from in vitro polarized human and murine Th2 cells, and also acts as a Th2 chemotactic factor. IL-33- promoting cytokine production activates human basophils and probably also regulates their migration. IL-33 influence on activation, degranulation, enhanced adhesion and survival of eosinophils has been shown. Mast cells are well-studied responders to IL-33. The cytokine increases synthesis of IL-6, IL-13, IL-1β, TNF-α, prostaglandin D2 and MCP-1 by primary bone marrow-derived mast cells. Other cells activated by IL-33 are cardiomyocytes, glial cells and CD34+ cells.
Find documents for the lot
Example Instructions for Use (RUO)
Example Instructions for Use (RUO)
Safety Information (RUO)
MSDS (RUO)