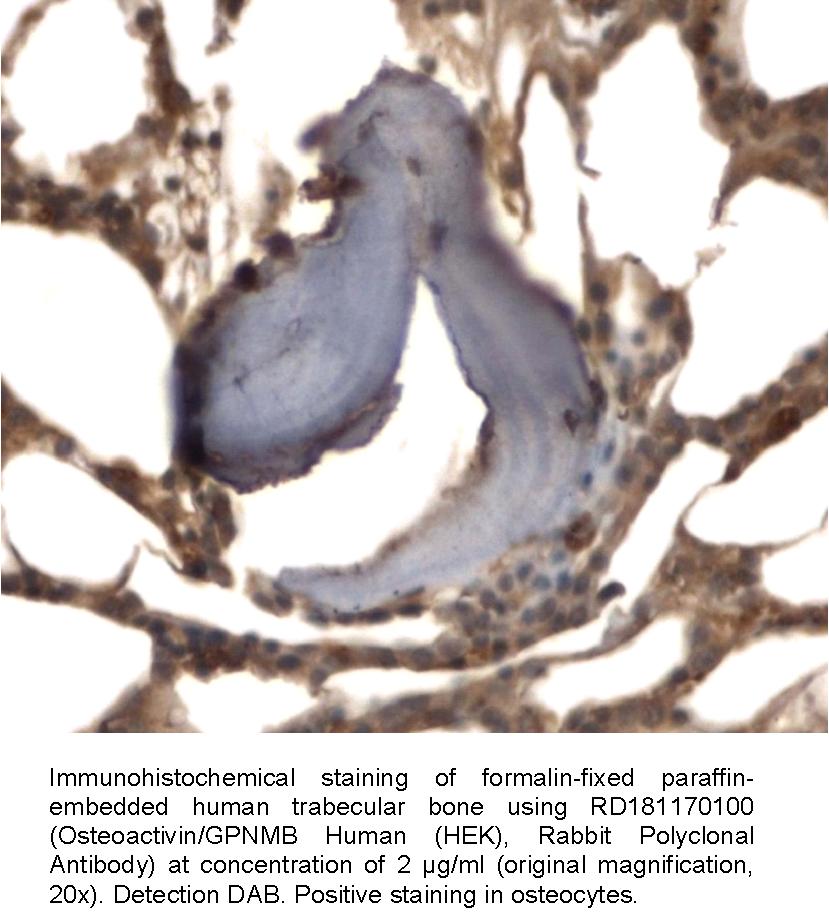
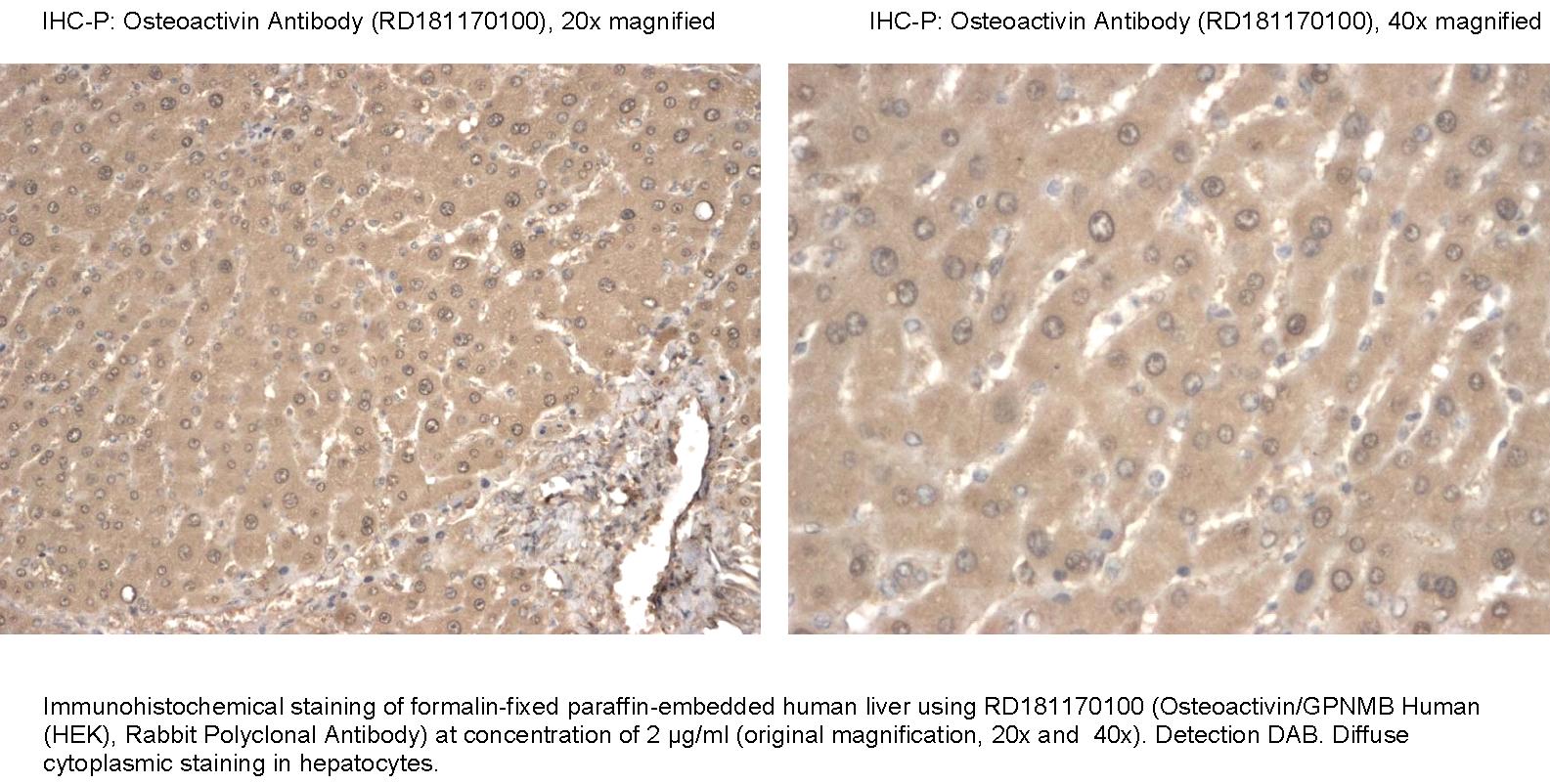
Type
Polyclonal Antibody
Applications
Immunohistochemistry
Antibodies Applications
Source of Antigen
HEK293
Hosts
Rabbit
Preparation
The antibody was raised in rabbits by immunization with the recombinant Human Osteoactivin.
Amino Acid Sequence
Total 477 AA (whole extracellular domain Ala 22 – Pro 486). MW: 53.7 kDa (calculated), migrates at ~ 97 kDa on SDS-PAGE; C-terminal linker (Lys, Leu) and 10× His-tag (10 extra AA).
AKRFHDVLGNERPSAYMREHNQLNGWSSDENDWNEKLYPVWKRGDMRWKNSWKGGRVQAVLTSDSPALVGSNITFAVNLIFPRCQKEDANGNIVYEKNCRNEAGLSADPYVYNWTAWSEDSDGENGTGQSHHNVFPDGKPFPHHPGWRRWNFIYVFHTLGQYFQKLGRCSVRVSVNTANVTLGPQLMEVTVYRRHGRAYVPIAQVKDVYVVTDQIPVFVTMFQKNDRNSSDETFLKDLPIMFDVLIHDPSHFLNYSTINYKWSFGDNTGLFVSTNHTVNHTYVLNGTFSLNLTVKAAAPGPCPPPPPPPRPSKPTPSLATTLKSYDSNTPGPAGDNPLELSRIPDENCQINRYGHFQATITIVEGILEVNIIQMTDVLMPVPWPESSLIDFVVTCQGSIPTEVCTIISDPTCEITQNTVCSPVDVDEMCLLTVRRTFNGSGTYCVNLTLGDDTSLALTSTLISVPKLHHHHHHHHHH
Species Reactivity
Human. Not yet tested in other species.
Purification Method
Immunoaffinity chromatography on a column with immobilized recombinant Human Osteoactivin.
Antibody Content
0.1 mg (determined by BCA method, BSA was used as a standard)
Formulation
The antibody is lyophilized in 0.05 M phosphate buffer, 0.1 M NaCl, pH 7.2.
Reconstitution
Add 0.2 ml of deionized water and let the lyophilized pellet dissolve completely. Slight turbidity may occur after reconstitution, which does not affect activity of the antibody. In this case clarify the solution by centrifugation.
Shipping
At ambient temperature. Upon receipt, store the product at the temperature recommended below.
Storage/Expiration
The lyophilized antibody remains stable and fully active until the expiry date when stored at -20°C. Aliquot the product after reconstitution to avoid repeated freezing/thawing cycles and store frozen at -80°C. Reconstituted antibody can be stored at 4°C for a limited period of time; it does not show decline in activity after one week at 4°C.
Quality Control Test
Indirect ELISA – to determine titer of the antibody
SDS PAGE – to determine purity of the antibody
BCA - to determine quantity of the antibody
Note
This product is for research use only.
Research topic
Bone and cartilage metabolism, Neural tissue markers, Oncology, Renal disease
Summary
Osteoactivin (OA), also known as Dchil (dendritic cell-associated, heparin sulfate proteoglycan-dependent integrin ligand), Gpnmb (glycoprotein non-metastatic melanomal protein B), or Hgfin (hematopoietic growth factor-inducible neurokinin 1), is a transmembrane glycoprotein. The OA gene, located on human chromosome 7p15.1 encodes a protein of 572 amino acid residues. OA may exist as a 65-kD unglycosylated cellular protein or as multiple glycosylated proteins with molecular size varying between 80-kD to 139-kD. Glycosylation of proteins plays a crucial role in cell differentiation and function. The transmembrane OA can be proteolytically cleaved by extracellular proteases, such as ADAMs and MMPs, in a process called ectodomain shedding, which results in the detachment and release of the extracellular domains which act as cytokines or growth factors. OA is expressed in a wide array of tissues and plays a regulatory role in various cellular functions. OA expression is associated with cell differentiation with high expression levels of OA protein found in the nervous system, basal layer of the skin, germinal cells of hair follicles, and in developing nephrons of the kidney in mouse embryos. In immune cells, OA was detected in differentiated macrophages, lymphocytes, and dendritic cells, but undetectable in proliferating hematopoietic progenitors. It was reported that osteoblast-derived OA has a regulatory role in osteoblast differentiation and bone formation. OA expression in osteoblasts is up-regulated by bone morphogenetic protein-2 (BMP-2) and OA appears to be a key mediator of BMP-2-induced osteoblast differentiation. Because of its suggested functions in cell adhesion, migration, and differentiation in various cell types and tissues, OA has been implicated in physiological and pathophysiological cascades of tissue injury and repair. In addition to its diverse roles in normal cells and tissues, aberrant OA expression is linked to various pathological disorders such as glaucoma, kidney disease, osteoarthritis, amyotrophic lateral sclerosis and several types of cancer, including: uveal melanoma, glioma, hepatocellular carcinoma, and cutaneous melanoma. The discovery that osteoactivin is selectively overexpressed in aggressive bone metastatic breast cancer cells suggests an important role for this molecule in the progression to metastatic breast cancer.