Type
Sandwich ELISA, HRP-labelled antibody
Applications
Serum, Cell culture supernatant
Sample Requirements
10 µl/well
Shipping
At ambient temperature. Upon receipt, store the product at the temperature recommended below.
Storage/Expiration
Store the complete kit at 2–8°C. Under these conditions, the kit is stable until the expiration date (see label on the box).
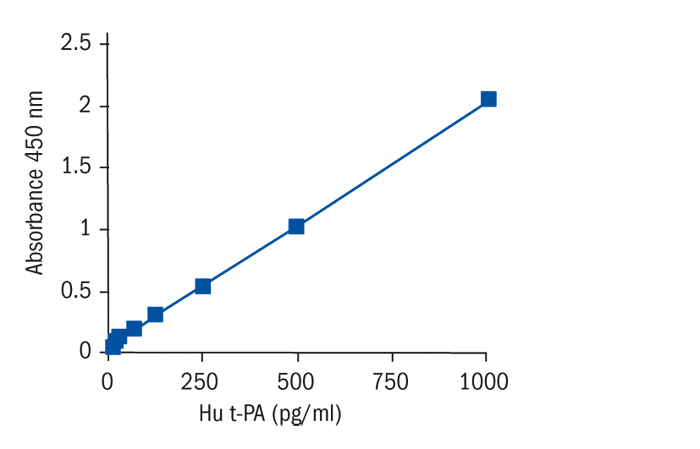
Calibration Curve
Calibration Range
15.6–1000 pg/ml
Limit of Detection
6.4 pg/ml
Intra-assay (Within-Run)
CV = 3.6%
Inter-assay (Run-to-Run)
CV = 6.5%
Spiking Recovery
98,00%
Dilution Linearity
100,00%
Research topic
Others
Summary
Tissue-type plasminogen activator (t-PA) is a serine protease which occurs in blood plasma, serum, other body fluids, tissues and conditioned media of certain cultured cells. It can convert the inactive proenzyme plasminogen to the active protease plasmin. Plasmin can degrade fibrin, the matrix of a blood clot in a process known as fibrinolysis, leading to dissolution of the clot. Furthermore plasminogen activation is implicated in metastatic spread of malignant cells and in tissue remodelling.
Fibrin has been shown to accelerate the conversion of plasminogen to plasmin which is mediated by t-PA. Through this pathway fibrin promotes its own degradation. Inhibitors to t-PA have been found in blood preparations, cell culture media and tissues. These plasminogen activator inhibitors -1 and -2 (PAI-1, PAI-2) react extremely rapidly with t-PA, forming inactive complexes. The availability of free active t-PA is regulated through this interaction. A correlation between low serum levels of t-PA activity and thrombotic tendency has been
described. An impaired release of t-PA from the endothelium in Graves‘ disease with significantly lowered
basal plasma t-PA levels was described. The clinical evaluation of t-PA levels in patients with liver diseases revealed a change of the t-PA levels in the clinical causes of these pathologies with increased t-PA levels with progression of the liver disease. Elevated levels of t-PA in serum were shown to occur in relation to retinopathy in type 1 diabetes mellitus.
The major field of clinical interest is the field of diseases of the heart. t-PA plasma levels have been shown to be altered with the presence of transplant coronary artery disease in cardiac transplant recipients. t-PA is described as a factor correlating to the risk of development of cardiovascular disease as was shown for controls and long-term dialysis patients. Changes of t-PA levels were shown in myocardial infarction. In stroke patients, high t-PA antigen concentrations indicate an activation of the fibrinolytic system or a complex formation with the inhibitors.
Find documents for the lot
Example Instructions for Use (RUO)
Example Instructions for Use (RUO)
Safety Information (RUO)
MSDS (RUO)