Update 2026: miREIA assay kits have been discontinued as of January 1, 2026.
The microRNA project now continues exclusively on the Two-Tailed RT-qPCR (TT-PCR) platform.
Extracellular vesicles are small extracellular membrane-enclosed vesicles with a lipid bilayer membrane containing different lipids, metabolites, proteins, RNAs and DNAs. Although discovered as early as 1967 and considered to be cell debris they were later recognized as bioactive mediators in cell-to -cell communication and critical players in both physiological and pathological conditions in the 1990s.
Different types of cells produce extracellular vesicles with different contents due to distinctive cues from their environment. Quantifying and identifying the molecules in the extracellular vesicles (EVs) is challenging. In response to certain conditions, such as stress or injury, the molecules may be up- or down- regulated, presenting a unique signature that could serve as a potential diagnostic or prognostic biomarker for cardiovascular diseases.
Various species of RNA have been identified in extracellular vesicles, including mRNA, tRNA, small interfering RNA (siRNA), long-non-coding RNA (lncRNA) and microRNA (miRNA). Total RNA sequencing of extracellular vesicles from human serum revealed that tRNA and miRNA made up approximately 15% of RNA. Among identified RNA, miRNA has been closely associated with cardiovascular diseases.
Many different miRNAs, such as miR-21, miR-126 and miR-146a, have been implicated in the pathogenesis of different stages of cardiovascular diseases, including atherosclerosis, cardiac hypertrophy, myocardial infarction and heart failure.
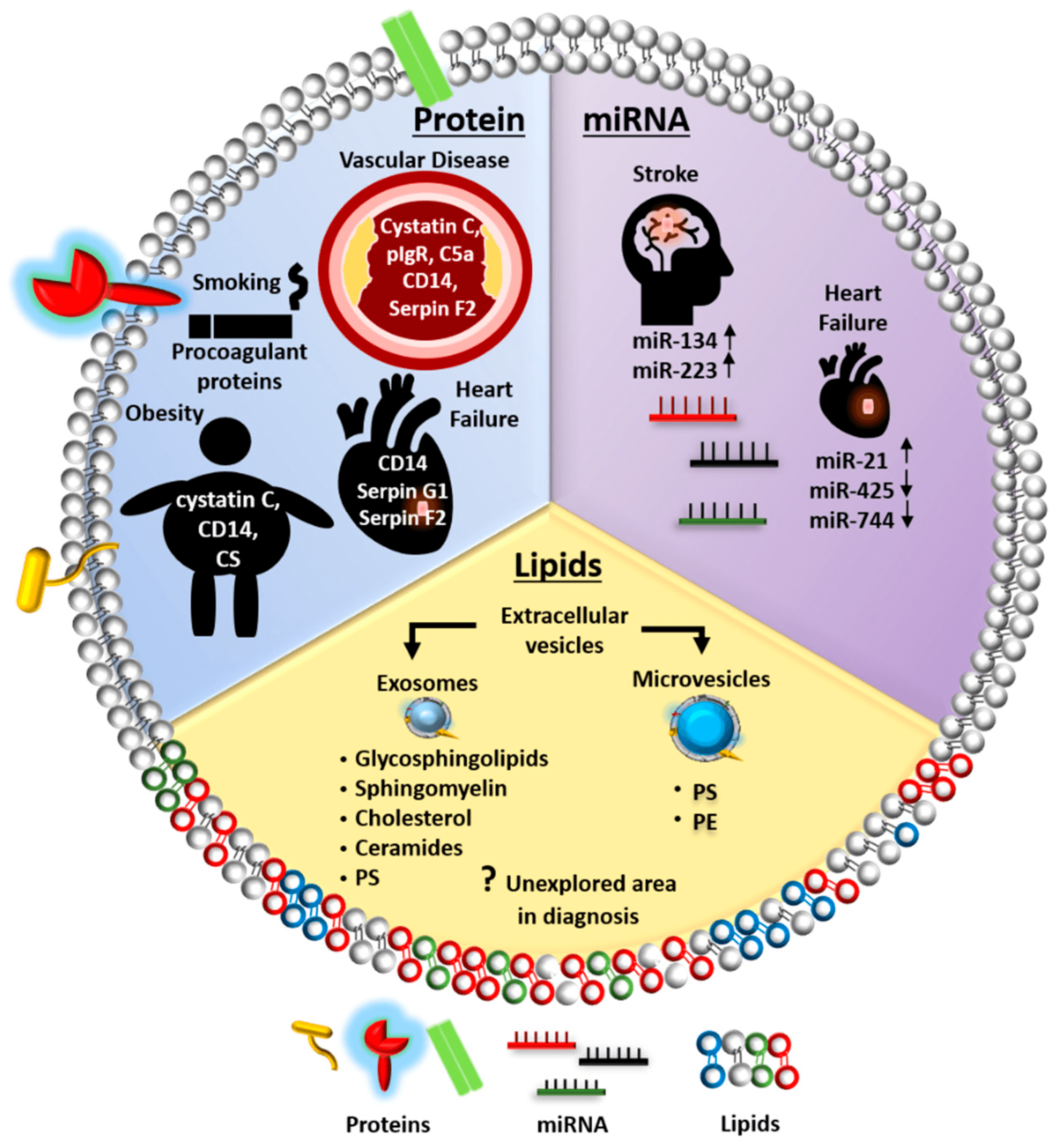
In acute ischemic stroke patients, higher levels of miR-134 and miR-223 from extracellular vesicles correlated with National Institutes of Health Stroke Scale scores and brain infarct volume and, consequently, with a worse prognosis.
In heart failure patients, higher levels of miR-2, and lower levels of miR-425 and miR-744 in extracellular vesicles were reported.
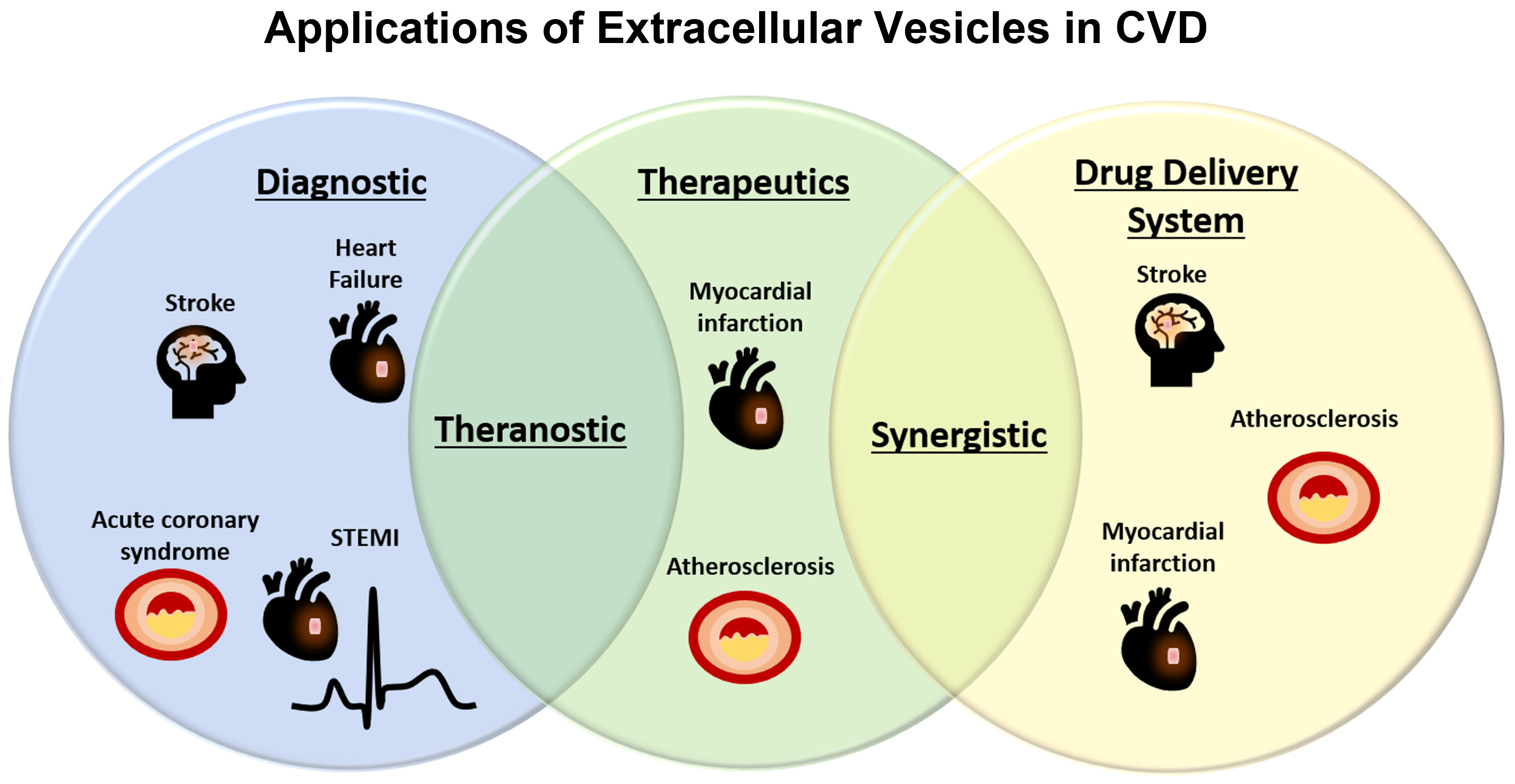
Extracellular vesicles miRNAs as biomarkers in cardiovascular disease
The utility of extracellular vesicles miRNAs as biomarkers in predicting the risk of developing cardiovascular disease in individuals with high-risk factors, such as exposure to smoke, obesity, and diabetes, has also been explored. Alteration of extracellular vesicles miRNA upon exposure to particulate matter correlates with increased blood pressure and coagulant state. Some miRNAs have been proposed as early biomarkers to predict cardiovascular events in obese patients.
More challenges for extracellular vesicles and miRNAs
Extracellular vesicles miRNAs are studied not only as new diagnostic markers, but also as therapeutic agents and a potential drug delivery system in cardiovascular diseases.
In several cell-based therapy preclinical studies and clinical trials only limited transplanted cells differentiated into new cardiomyocytes. As a most promising alternative a bioactivity of stem cell-derived extracellular vesicles was described which enables trigger endogenous reparation mechanisms.
Extracellular vesicles derived from cardiomyocytes instead of having therapeutic effects may convey under pathophysiological conditions “danger signals” to other cells. In contrast, extracellular vesicles released from cardiac progenitor cells, are mostly beneficial, regardless of the microenvironment they are generated in. These extracellular vesicles promoted angiogenesis, were anti-fibrotic, and their administration in vivo was able to reduce infarct size and improved heart function in animals subjected to myocardial infarction.
The intrinsic therapeutic activity and cell-to-cell communication roles of extracellular vesicles have led to of the idea of exploiting these vesicles as a drug delivery system for targeted release of bioactive molecules. Extracellular vesicles as an endogenous counterpart to liposomes may offer an opportunity to overcome some of liposomes’ limitations.
An in vitro study on atherosclerosis demonstrated the potential extracellular vesicles of EVs in delivering miRNAs into primary endothelial cells isolated from the aorta. As atheroprotective miRNAs, miR-143, miR-145 and miR-126 have been identified and used in several therapeutic studies.
Intravenous administration of EVs loaded with miR-93-5p reduced myocardial damage and infarct size. Similar treatment efficacy was found from vesicles with miR-126 or miR-146a.
Ischemic stroke has been studied in the rodent stroke model. Intravenous administration of extracellular vesicles containing miR-17-92 significantly reduced neurological deficit. It was also confirmed that extracellular vesicles can cross the blood-brain-barrier, the main impediment of drug delivery systems. This finding predestines extracellular vesicles as a novel and attractive drug delivery system.
